Opportunities for Collaboration with the Trial Innovation Network
If you’re doing multi-site clinical research, the Trial Innovation Network and its partner the Recruitment Innovation Center can supercharge your study recruitment and make technical details such as trial protocol and data management easier and less time-consuming. True to its name, this is a collaborative project with teams of experts across CTSA Program Hubs who comprise both Trial Innovation Centers (TICs) and the Recruitment Innovation Center (RIC).
Meet the Trial Innovation Network
TICs and RIC and not only fun acronyms to say, but they are helpful teams of people who have dedicated their efforts to improving collaboration among CTSAs as well as improving the actual trials themselves.
As you might expect, Trial Innovation Centers specialize in trial protocol, budgets, single IRB, data management, and other technical aspects of the study.
The Recruitment Innovation Center is focused on the vast array of recruitment efforts including cohort discovery, community engagement studios, and planning recruitment methods.
The Trial Innovation Network is not only the TICs and RIC, but they work with the CTSA Program Hubs, NCATS, NIH ICs, FDA, PCORI, participants, providers, the public, and industry.
Click here to view and/or download the flyer as a pdf.
Free Resources
Anyone can benefit from the Trial Innovation Network’s free toolbox resources for investigators and research teams. Here’s a small sampling of what they currently offer, and if you have something you’d like to share, they’d love to grow this resource by adding more cool stuff from you, too!
The materials on this site are designed primarily for the CTSA Program staffs and institutions participating in the Trial Innovation Network. Other parties conducting engagement, recruitment, and retention activities for clinical trials and multi-site studies are welcomed to adopt these resources for local use as well. Those adopting the resources for their local use are expected to appropriately cite the resources used in the toolbox and give full credit to original developer, the contributor(s), as well as the Trial Innovation Network.
Topics include trial compensation, study websites, social media & digital recruitment, community engagement, and more.
Teams share expertise, methods and/or best practices to improve the way trials are conducted in these 45-min with 15-min Q&A sessions. Register for free, but don’t sweat it if you miss an interesting topic because all sessions are recorded and archived here as well.
Topics include recruitment, e-consent, collaboration, community, single IRB, online screening, return of value, and more.
How to Submit a Proposal for Network Support
If you have, or plan to have, a research proposal you think could benefit from additional feedback and support, find out more about what the Trial Innovation Network can do for your research.
Before you apply there are a few key things to know:
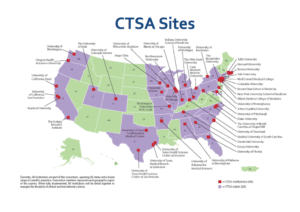
- You need to be affiliated with an institution that’s part of the CTSA Program. To find out if you are, check this map here.
- Collaboration is key. Studies requesting support must be multi-site.
- Be ready to explain the ways your research is “innovative.”
- Applications are rolling, and response time is five business days after submission.
- If a submission is not approved, recommendations are often made and resubmission is allowed.
- If a proposal is approved, communication generally begins soon after and continues on as needed in a very organized and efficient pattern.








0 Comments