If They Can’t Trust You with Stats, They Won’t Trust You with Money
For most grants you write, here is all you need to do to get them funded:
1. Convince me that it will be a crime against humanity if the science doesn’t get done, and that
2. No one in the world besides you can do it.

Simple.
For grants with a clinical trial, though, there is one more thing that you have to do, which seems simple when I say it, but people still screw it up continuously.
3. Get the statistics and recruitment right.
For clinical trials, (1) is usually pretty easy – by the time you’re proposing a clinical trial, you presumably have a mountain of preclinical evidence on the efficacy of whatever you’re proposing in a variety of animal models, you’ve probably got some safety data, and you’ve got a molecular mechanism that’s compelling. Nine out of ten reviewers are going to agree with you that it’s time to bring it into humans.
And then you blow it by setting up your trial design and statistics in such a way that you convince reviewers that no matter what happens, you’re not going to end with meaningful data. Your recruitment assumes literally everyone who comes through your clinic agrees to participate (or you don’t say how many people are coming through your clinic at all). Your recruitment requires you to be recruiting 24/7 – but you don’t even have a clinical coordinator, let alone a research nurse or two. Are you gonna sleep in the ER?
You power it based on wildly optimistic assumptions of efficacy – one clinical grant I recently reviewed required tenfold higher effectiveness than any other drug ever tested in order for detectable effect. You ignore effects of other treatments, or variability in the etiology of the underlying disease, in your inclusion/exclusion criteria (shock from blunt trauma ain’t the same as shock from burns and shock from infection). You include subgroup analyses you’re really not powered for. You include large numbers of secondary endpoints without telling me how you’re correcting for multiple testing (I like lots of endpoints, but you need to be careful with them statistically).
Your statistics section was obviously stolen from some other grant, so it has statistics-sounding words, that don’t make sense in current context.
Many authors don’t get power right:
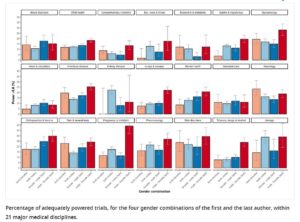
https://elifesciences.org/articles/34412/figures
Two big pieces of advice:
- If you’re not expert on it yourself, get a biostatistician involved in your trial design right from the start. Don’t just get someone to give you their CV to include.
- Get somebody who’s actually run a trial to help with the design and recruitment strategy. It’ll keep you from making the sorts of mistakes that call into question your ability to complete the study.
I just came off of a study section that was nothing but reviews of clinical trials. Essentially every one of them had these problems – and we all loved the premise and preliminary data on all of them. What that means is, get this piece right, and you’ve got a big jump on the competition.





0 Comments